 From the first confirmed US case of COVID-19 on Jan. 21st, 2020, to the “stay at home” order that was initiated in March, the fallout of the pandemic started to hit our economy. By May 2020, 38.6 million people had lost their jobs and unemployment rates rose to 14.7%. The largest unemployment rate since the great depression. It will take years to recover from the physical, psychological, and economic damage done to our country.
From the first confirmed US case of COVID-19 on Jan. 21st, 2020, to the “stay at home” order that was initiated in March, the fallout of the pandemic started to hit our economy. By May 2020, 38.6 million people had lost their jobs and unemployment rates rose to 14.7%. The largest unemployment rate since the great depression. It will take years to recover from the physical, psychological, and economic damage done to our country.
Many small and medium-sized businesses have had to shut down and layoff workers. However, for the pharmaceutical door manufacturers, COVID-19 has both helped and hindered their businesses. The pandemic has created a short-term burst of business which is positive, but supply chains and labor have been stretched thin.
Building a vaccine production facility takes years and costs billions of dollars. In a critical health emergency, manufacturers will seek to leverage existing infrastructure and form strategic partnerships rather than creating new buildings. The chief investments will be in equipment and process technology, not infrastructure. Finding the right door manufacture is crucial when it comes to the pharmaceutical and life science industry. Finding a partner that does not just sell doors but also offers a solution can go a long way. Building a trusted relationship will help solve challenges that come with retrofitting existing infrastructure.
Cleanroom Design
As suppliers to the life sciences communities, pharmaceutical door manufactures are on the front lines of the pandemic response. Vaccine production is a very complex process. Vaccine cleanrooms are built and customized to suit the unique process they require. While every vaccine approach is different, there are common cleanroom considerations that span the applications and technology platforms.
- Door interlocking and airlocks
- Traffic flow
- Process distribution
- Environmental monitoring
- Airflow segregation
- ISO classification, temperature & humidity
- Gowning and cleaning protocols
Common Vaccine Cleanroom Specification
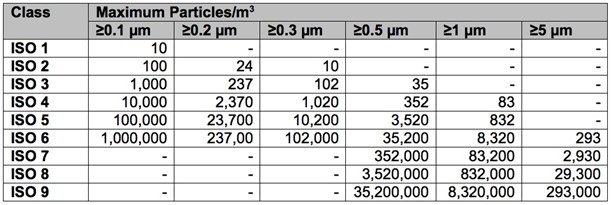
Cleanrooms are classified according to the cleanliness level of the air inside them. The International Organization for Standardization (ISO, is an independent, non-governmental organization that uses standards that aid in the creation of products and services that are safe, reliable, and are good quality. The cleanroom class is the level of cleanliness the room meets, according to the quantity and size of particles per volume of air. The primary authority in the US and Canada is the ISO classification system ISO 14644-1. ISO cleanroom classifications conform to the older U.S. Feder Standard 209, which is no longer in effect. However, it is still widely cited and can be easily cross-referenced with ISO classification. Below are the listings of the two classifications:
ISO 14644-1 Cleanroom Classifications
U.S. Federal Standard 209 Cleanroom Classifications
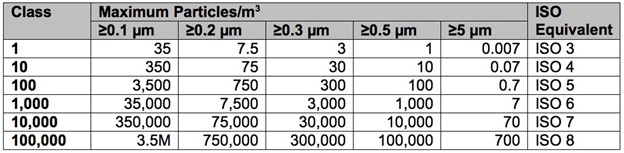
U.S. Federal Standard 209 Cleanroom Classifications
 When considering a certain ISO classification for a cleanroom, it is critically important to ensure the door manufacture capabilities align with the scope of the ISO level that is required. Vaccine cleanrooms are comprised of various ISO performance classifications ranging from ISO 4 (Class 10) to ISO 8 (Class 100,000). Specifications for vaccine cleanrooms vary based on process requirements.
When considering a certain ISO classification for a cleanroom, it is critically important to ensure the door manufacture capabilities align with the scope of the ISO level that is required. Vaccine cleanrooms are comprised of various ISO performance classifications ranging from ISO 4 (Class 10) to ISO 8 (Class 100,000). Specifications for vaccine cleanrooms vary based on process requirements.
Regulatory Considerations
In addition to the ISO requirements, there are a few other regulatory considerations. We have detailed them below:
Good Manufacturing Practices (GMP) are the practices required to conform to the guidelines recommended by agencies that control the authorization and licensing of the manufacture and sales of food and beverages, cosmetics, pharmaceutical products, dietary supplements, and medical devices. These guidelines provide minimum requirements that a manufacturer must meet to assure they consistently provide high-quality products. The rules that govern each industry may differ significantly, however, the main purpose of GMP is always to prevent harm from occurring to the end-user.
US Food and Drug Administration (FDA) is responsible for protecting public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological products, and medical devices.
European Medicines Evaluation Agency (EMEA) is an agency of the European Union in charge of the evaluation and supervision of medicinal products. Its main responsibility is the protection and promotion of public and animal health.
Cleanroom Doors
Cleanroom doors are just one of the materials that go into the design and construction of a cleanroom. These doors need to meet these strict GMP, ISO, FDA, EMEA, and cleanroom requirements. With various door material options, most choose seamless molded fiberglass doors. These are non-porous, non-shedding, non-absorptive, and ideal for sterile environments. These doors can be engineered with multiple options including an interlock access control system to maintain cleanroom pressure differentials as well as keep contaminants out.
So where does this leave us?
- Efforts to increase efficiencies will continue to improve.
- The global response to the pandemic will likely go on for years.
- Manufacturers will continue to improve and expand their facilities.
- Vaccine manufacturers, large and small will find new ways to partner with other life science companies.
- All of this will continue to provide ongoing opportunities for pharmaceutical equipment manufacturers.
The Coronavirus pandemic exposed massive gaps in US preparedness at many levels. However, the US continues to be the leader in development and innovation in the life science industries at all levels. Bioengineering, Gene Therapy, Cell Therapy, and many more new discoveries will continue to drive innovation and expansion into the future.
ASI Doors has been in the industry for over 50 years and has become the leader in cleanroom door technology. With our vast experience, we help make sure you meet all the requirements necessary to meet today’s challenges. Contact us today!
